2013
Teerawut Bootwicha, Xiangqian Liu, Roman Pluta, Iuliana Atodiresei, Magnus Rueping
Angew. Chem. Int. Ed. 2013, 52, 12856-12859
ABSTRACT:
Cinchona alkaloid catalysts in combination with air- and moisture-stable N-trifluoromethylthiophthalimide as electrophilic SCF3 source enabled the catalytic enantioselective trifluoromethylsulfenylation. Thus, a series of α-SCF3 esters that bear a quaternary carbon stereogenic center were obtained with excellent yield and enantioselectivity. Moreover, the products can be readily converted into valuable α-SCF3 β-hydroxyesters. We have developed a highly enantioselective cinchona alkaloid catalyzed trifluoromethylsulfenylation of β-ketoesters with N-trifluoromethylthiophthalimide as electrophilic SCF3 source. This enantioselective method enables the construction of a quaternary carbon stereocenter that bears a SCF3 group. In general, the products are obtained in good yields with excellent enantioselectivities. Depending on the employed alkaloid quinine or quinidine, either the (R)- or (S)-configured product can be obtained. Furthermore, the highly diastereoselective addition of Grignard reagents allows the formation of the corresponding α-SCF3 β-hydroxyesters in high yields.
Erli Sugiono and Magnus Rueping
Beilstein J. Org. Chem. 2013, 9, 2457–2462
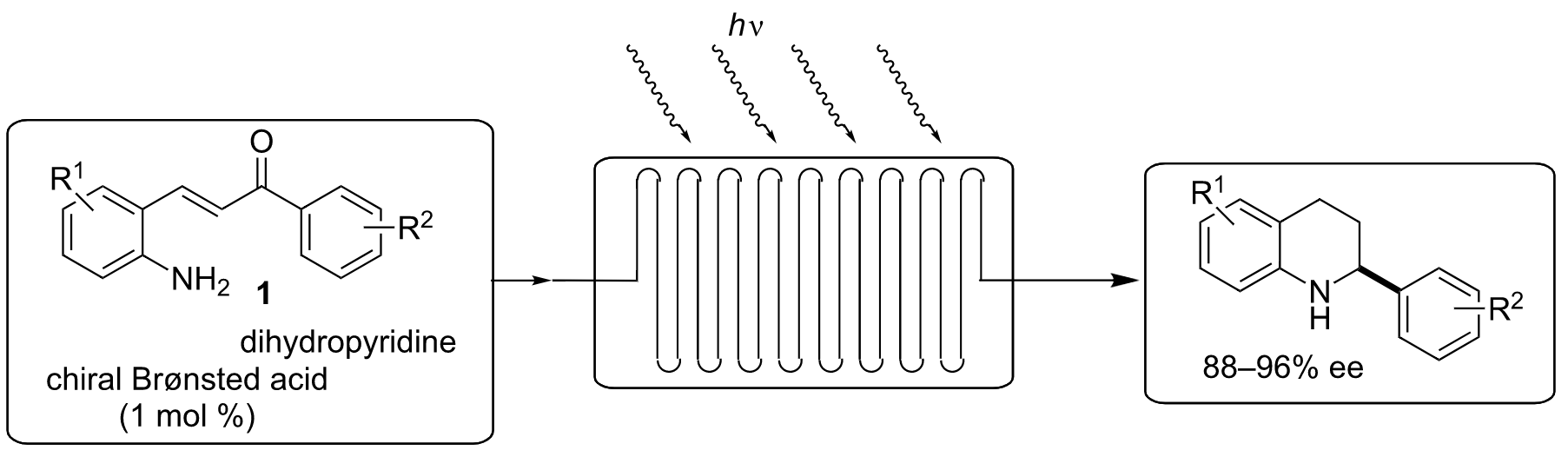
ABSTRACT:
A continuous-flow asymmetric organocatalytic photocyclization–transfer hydrogenation cascade reaction has been developed. The new protocol allows the synthesis of tetrahydroquinolines from readily available 2-aminochalcones using a combination of photochemistry and asymmetric Brønsted acid catalysis. The photocylization and subsequent reduction was performed with catalytic amount of chiral BINOL derived phosphoric acid diester and Hantzsch dihydropyridine as hydrogen source providing the desired products in good yields and with excellent enantioselectivities. We have demonstrated the great potential of a new continuous-flow microreactor system for the photocyclization–reduction cascade of 2-aminochalcones. Under the continuous-flow condition a variety of substituted 2-aminochalcones underwent the photocyclization and the subsequent transfer hydrogenation to afford a series of differently substituted tetrahydroquinolines in good yields and with excellent enantioselectivities. This efficient protocol for the synthesis of tetrahydroquinoline from readily available 2-aminochalcone provides an attractive alternative to the existing procedures and serves as a basis for further exploration of this new concept.
Karl Kaupmees, Nikita Tolstoluzhsky, Sadiya Raja, Magnus Rueping, Ivo Leito
Angew. Chem. Int. Ed. 2013, 52, 11569-11572
ABSTRACT:
pKa values have been determined for a series of chiral Brønsted acids in AN on the basis of a UV/Vis spectrophotometric method. For the three groups of catalysts investigated, it can be concluded that the binol-derived phosphoric acids that make up group A have a pKa range of 12–14, the fluorinated N-sulfonylphosphoramides in group B have acidity values around 6–7 on the pKa scale, and bis(sulfuryl)imides (group C) have pKa values of approximately 5 in AN. An acidity scale was compiled that includes various other known acids as well as the chiral Brønsted acids and will be useful for the further development of the field of asymmetric Brønsted acid catalysis. There is a direct correlation of the catalytic properties of these acids with their pKa values, whereby higher rate constants are observed for more acidic Brønsted acid catalysts. It is also evident from the present study that the big gap in acidity between N-triflylphosphoramides and phosphoric acids calls for the development of new chiral catalysts with adjusted catalytic properties.
Magnus Rueping, Nikita Tolstoluzhsky, Pavlo Nikolaienko
Chem. Eur. J. 2013, 19, 14043-14046
ABSTRACT:
A new and general method has been developed for the synthesis of vinyl trifluoromethyl thioethers starting from readily available vinyl iodides. A wide variety of differently substituted alkyl and aryl di-, tri-, and tetrasubstituted substrates were applied in the reaction with CuSCF3 to give the corresponding products in high yields within short reaction times. Notably, the initial E/Z isomer ratio was retained after the reaction. In addition, a new copper-catalyzed trifluoromethyl thioetherification protocol has been developed. The procedure makes use of the [Bu4N][SCF3] salt and provides the trifluormethyl thioether in good yields. In contrast to previous methods the protocols developed show broad scope, use safe and non-toxic metals and reagents, and can be carried out under mild reaction conditions, making them useful for organic synthesis.
Quentin Lefebvre, Marc Jentsch and Magnus Rueping
Beilstein J. Org. Chem. 2013, 9, 1883–1890
ABSTRACT:
A continuous flow oxidative photocyclization of stilbene derivatives has been developed which allows the scalable synthesis of backbone functionalized phenanthrenes and helicenes of various sizes in good yields. We have developed a new photo-flow methodology [28-32] for the synthesis of phenanthrenes and helicenes. Although photocyclization of stilbene derivatives was disclosed more than 40 years ago, this is the first report of UV-light-driven photocyclization in flow. In general phenantrenes as well as [4]-, [5]- and [6]helicenes with different substitution patterns are obtained in good to excellent yields. In addition our first attempts to scale up the flow photocyclization reactions were successful providing the opportunity for multi-gram syntheses.
Arindam Das, Chandra M. R. Volla, Iuliana Atodiresei, Wolfgang Bettray, Magnus Rueping
Angew. Chem. Int. Ed. 2013, 52, 8008-8011
ABSTRACT:
The work described herein represents the first report on the catalytic asymmetric 6π electrocyclization reaction for the synthesis of enantiomerically enriched 1,4-dihydropyridazines. The protocol allows the synthesis of various valuable 1,4-dihydropyridazine derivatives with an excellent level of enantioselectivity. Readily available substrates, operational simplicity, as well as high tolerance toward functional groups are features of this newly developed procedure. Given the difficulties associated with the development of asymmetric electrocyclization reactions, this new asymmetric Brønsted acid catalysis procedure provides a good basis and scope for further extensions and explorations. The work described herein represents the first report on the catalytic asymmetric 6π electrocyclization reaction for the synthesis of enantiomerically enriched 1,4-dihydropyridazines. The protocol allows the synthesis of various valuable 1,4-dihydropyridazine derivatives with an excellent level of enantioselectivity. Readily available substrates, operational simplicity, as well as high tolerance toward functional groups are features of this newly developed procedure. Given the difficulties associated with the development of asymmetric electrocyclization reactions, this new asymmetric Brønsted acid catalysis procedure provides a good basis and scope for further extensions and explorations.
Chien‐Chi Hsiao, Hsuan‐Hung Liao, Erli Sugiono, Iuliana Atodiresei, Magnus Rueping
Chem. Eur. J. 2013, 19, 9775-9779
ABSTRACT:
We describe a new light driven asymmetric ion-pair catalysis procedure for performing an enantioselective hydrogenation of pyrylium ions. The newly developed dual and combined photo-assisted Brønsted acid catalyzed procedure has broad scope and allows, for the first time, access to valuable 4H-chromenes in good yields and with excellent enantioselectivities. The reaction sequence consists of a dual light and Brønsted acid mediated isomerization–cyclization reaction to yield chroman-2-ol intermediates. The subsequent Brønsted acid catalyzed elimination of water leads to an unprecedented intermediary chiral ion pair consisting of a benzopyrylium ion and a chiral phosphate anion. The following organo-hydride addition, exclusively occurring in the 4-position, provides the desired enantioenriched 4H-chromenes.Detailed mechanistic investigations demonstrate an unexpected synergistic effect as both light and the Brønsted acid catalyst are involved in both the cyclization and transfer hydrogenation events. In addition, the enantiodifferentiating fluorescence quenching has not been observed for BINOL-phosphoric acid derivatives, which make their use in light driven catalysis promising. From a synthetic view, the present approach is particularly attractive as it utilises readily available chalcones and avoids the preparation of rather sensitive and unstable chroman-2-ol intermediates. Thus, we are confident that the concept of asymmetric pyrylium ion pair catalysis, in which the chiral information is efficiently transferred from the Brønsted acid anion to the product, will find broad application.
Magnus Rueping, Carlos Vila, Teerawut Bootwicha
ACS Catal. 2013, 3, 1676-1680
ABSTRACT:
A continuous flow procedure for the efficient metal-free, visible light photoredox-catalyzed α-functionalization of tertiary amines has been developed. Rose Bengal has been identified as an effective organic photocatalyst for continuous flow C–C and C–P bond formations as well as multicomponent reactions. We have developed an environmentally acceptable, metal-free, photo-organocatalytic, continuous flow methodology that has been successfully applied in the α-functionalization of tertiary amines. Nitroalkanes, TMSCN, dialkyl malonates, and dialkyl phosphites were reacted with various N-aryl tetrahydroisoquinolines to provide the corresponding products in moderate to excellent yields. Furthermore, N,N-dimethylanilines were successfully reacted with different isocyanides in the Ugi-multicomponent reaction in flow, resulting in highly valuable α-amino amides in good yields. However, most importantly, considerably shorter reaction times are necessary if compared with the batch conditions. This is important for further reaction design. In addition, Rose Bengal is an inexpensive, readily available, environmentally benign organic dye that can be used as an efficient visible light photoredox catalyst in continuous flow fashion.
5. Visible Light Photoredox-Catalyzed Multi-Component Reactions
Magnus Rueping, Carlos Vila
Org. Lett. 2013, 15, 2092-2095
ABSTRACT:
Oxidative three-component reactions for the direct synthesis of α-amino amides and imides from tertiary amines have been developed. These reactions involve the functionalization of C(sp3)–H bonds adjacent to nitrogen atoms via mild aerobic oxidation using visible light photoredox catalysis. The protocols are applicable to a wide range of amines and isocyanides, as well as water and carboxylic acids, providing straightforward access to a variety of highly functionalized α-amino amides and imides. We have developed an oxidative three-component reaction for the synthesis of valuable α-amino amides and imides from tertiary amines, isocyanides, and water or carboxylic acids using visible light photoredox catalysis. The reaction proceeds smoothly using only 1 mol % of iridium photoredox catalyst and visible light without the need for an additional external oxidant. In general, the catalysis procedure tolerates various functional groups and can be conducted in open reaction vessels in the presence of air and moisture, providing a series of differently substituted products. Further investigations of the aerobic visible light photoredox catalysis to enable multicomponent reactions will be of relevance for further developing the field of photoredox catalysis.
Shaoqun Zhu, Arindam Das, Lan Bui, Hanjun Zhou, Dennis P. Curran, Magnus Rueping
J. Am. Chem. Soc. 2013, 135, 1823-1829
ABSTRACT:
Visible light photoredox catalyzed inter- and intramolecular C–H functionalization reactions of tertiary amines have been developed. Oxygen was found to act as chemical switch to trigger two different reaction pathways and to obtain two different types of products from the same starting material. In the absence of oxygen, the intermolecular addition of N,N-dimethyl-anilines to electron-deficient alkenes provided γ-amino nitriles in good to high yields. In the presence of oxygen, a radical addition/cyclization reaction occurred which resulted in the formation of tetrahydroquinoline derivatives in good yields under mild reaction conditions. The intramolecular version of the radical addition led to the unexpected formation of indole-3-carboxaldehyde derivatives. Mechanistic investigations of this reaction cascade uncovered a new photoredox catalyzed C–C bond cleavage reaction. We have developed a direct intermolecular and intramolecular C–H functionalization of tertiary amines by employing visible light photoredox catalysis. Notably, oxygen can be used as a chemical switch to trigger two different reaction pathways and to obtain two different types of products from the same staring material. On one hand, γ-amino nitriles were obtained in good to high yields in the absence of oxygen via an intermolecular addition of N,N-dimethyl-anilines to benzylidene malononitriles. On the other hand, in the presence of oxygen, a radical addition/cyclization reaction sequence occurred, leading to the corresponding tetrahydroquinoline derivatives in good yields, under mild reaction conditions. In addition, an intramolecular version has been developed. In this case, a photoredox catalyzed domino reaction involving C–C bond formation, aromatization, and carbon–carbon bond cleavage took place and afforded biologically and medicinally important precursor indole-3-carbaldehyde derivatives via the formation of α-amino alkyl radical intermediates. Mechanistic investigations have been performed to elucidate the reactive intermediates and to propose a plausible reaction pathway for the photoredox catalyzed addition/aromatization/C–C bond cleavage cascade reaction. Importantly, a new photoredox catalyzed C–C bond cleavage of indolylpropanones into the corresponding indolecarbaldehydes has been uncovered which in its more elaborated way holds great potential in organic synthesis. This catalysis is mechanistically new and can be further explored to design new transformations in the field of photoredox catalysis.
Carlos Vila and Magnus Rueping
Green Chem. 2013, 15, 2056-2059
ABSTRACT:
Visible-light mediated heterogeneous C–H functionalization of tertiary amines provides access to a variety of α-amino amides. An oxidative, titanium dioxide catalyzed, Ugi-type, three-component reaction has been developed in which the catalyst can be recycled without loss of activity. we have developed a new protocol for the synthesis of α-amino amides driven by visible-light catalysis and using cheap, robust, readily available and recyclable titanium dioxide. The advantage of using such a heterogeneous catalyst is the simplicity of its recycling and reuse. Thus, the protocol presented is both environmentally acceptable and economical. Further investigation into the applicability of this methodology for other multi-component reactions is currently ongoing and will be disclosed in due course.
Beata Kolesinska, Dominika J. Podwysocka, Magnus Rueping, Dieter Seebach, Faustin Kamena, Peter Walde, Markus Sauer, Barbara Windschiegl, Mira Meyer‐Ács, Marc Vor der Brüggen, Sebastian Giehring
Chemistry & Biodiversity 2013, 10, 1-38
ABSTRACT:
After a survey of the special role, which the amino acid proline plays in the chemistry of life, the cell-penetrating properties of polycationic proline-containing peptides are discussed. The fluorescein-labeled tetradecaproline is slowly taken up by rat kidney cells (NRK-49F). Here, we describe detailed observation that a hexa-β3-Pro derivative penetrates fibroblast cells, and we present the results of an extensive investigation of oligo-L- and oligo-D-α-prolines, as well as of oligo-β2h- and oligo-β3h-prolines without and with fluorescence labels (1–8). Permeation through protein-free phospholipid bilayers is detected with the nanoFAST biochip technology . This methodology is applied for the first time for quantitative determination of translocation rates of cell-penetrating peptides (CPPs) across lipid bilayers. Cell penetration is observed with mouse (3T3) and human foreskin fibroblasts (HFF). The stabilities of oligoprolines in heparin-stabilized human plasma increase with decreasing chain lengths. Time- and solvent-dependent CD spectra of most of the oligoprolines show changes that may be interpreted as arising from aggregation, and broadening of the NMR signals with time confirms this assumption.
Hsuan-Hung Liao, Chien-Chi Hsiao, Erli Sugiono, Magnus Rueping
Chem. Commun. 2013, 49, 7953-7955
ABSTRACT:
A new asymmetric photocyclization–reduction cascade employing readily available aminochalcones has been developed. The reaction sequence has been achieved by unifying photochemistry and asymmetric Brønsted acid catalysis and involves photocyclization followed by Brønsted acid catalyzed enantioselective hydrogenation in batch and flow. A highly enantioselective synthesis of differently substituted tetrahydroquinolines was accomplished via a new photocyclization–Brønsted acid catalyzed asymmetric reduction sequence of substituted 2-aminochalcones. Notably, a wide range of readily available 2-aminochalcones bearing substituted as well as unsubstituted aromatic and heteroaromatic residues on both ketone and enone moieties underwent photocyclization and subsequent asymmetric reduction to give products in good to high yields and with excellent enantioselectivities. The present protocol is environmentally friendly, allows a convenient, technically easy, metal-free, inexpensive synthesis of tetrahydroquinolines starting from readily available 2-aminochalcones and is an attractive alternative to existing procedures. Furthermore, the results demonstrate the feasibility of unifying photochemistry with asymmetric Brønsted acid catalysis in continuous flow chemistry, which is especially interesting for further exploration and scale up.